Percent yield = actual yield/ theoretical yield × 100. 
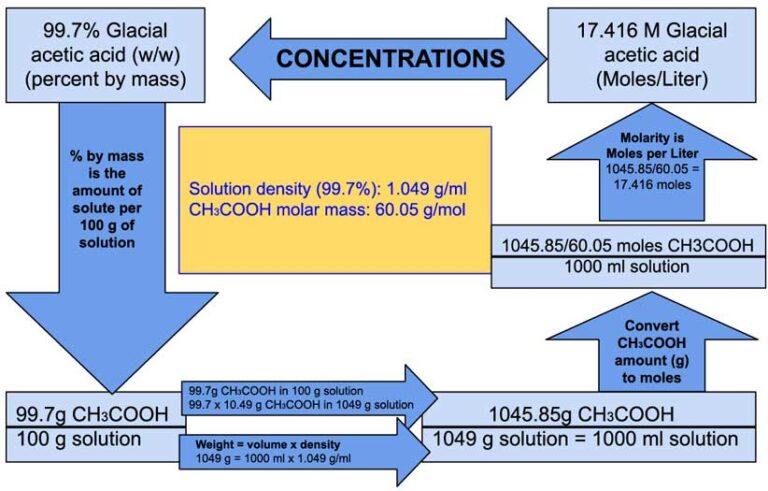
The actual yield is 1.0 g, theoretical yield = 1.19 grams Hence, the mass of isopentyl acetate= number of moles × molar mass= 0.0091745 × 130.19 grams per mol.įINAL STEP: calculating the percent yield. So, 0.0091745 mole isopentyl alcohol will produce 0.0091745 mol isopentyl acetate. By calculation, the molar mass of acetic acid comes out to be 60.05 g/mol. We can see that isopentyl alcohol is the limiting reagent. Acetic Acid CH3COOH - PubChem Apologies, we are having some trouble retrieving data from our servers. The molarity definition is based on the volume of. the solution is 1.67 M In case of acetic acid, 1 N solution 1 M solution Thus normality of 10 acetic acid is 1.67. Molarity (M)- is the molar concentration of a solution measured in moles of solute per liter of solution. Thus 1.67 moles of acetic acid is present in 1 L of water i.e. Acetic acid has the chemical formula CH 3 3 3COOH, which means it has two carbon atoms, four hydrogen atoms, and two oxygen atoms. So 100 g of acetic acid means 100/60 1.67 moles of acetic acid. => 1.05 grams of acetic acid/ 60.052 g/mol.įor isopentyl alcohol 0.809/ 88.179 grams per mol. 1 mole of acetic acid has a mass of 60 g. A dilute (approximately 5 percent by volume) solution of acetic acid produced by fermentation and oxidation of natural carbohydrates is called vinegar a salt, ester, or acylal of acetic acid is called acetate. Given - Molar mass of C6H10 82.2 g/mol Molar mass of O2 32 g/mol Molar mass of CO2 44g/mol Molar. Hence, for acetic acid the number of moles = mass/molar mass. acetic acid (CH3COOH), also called ethanoic acid, the most important of the carboxylic acids. This is the chemical formula for acetic acid (the chemical that gives the sharp taste to vinegar): CH,CO,H An analytical chemist has determined by measurements that there are 43.6 moles of carbon in a sample of acetic acid. This is done by converting the mass into moles. Since one mole of each reactants formed the product, we have to find the limiting reagent. 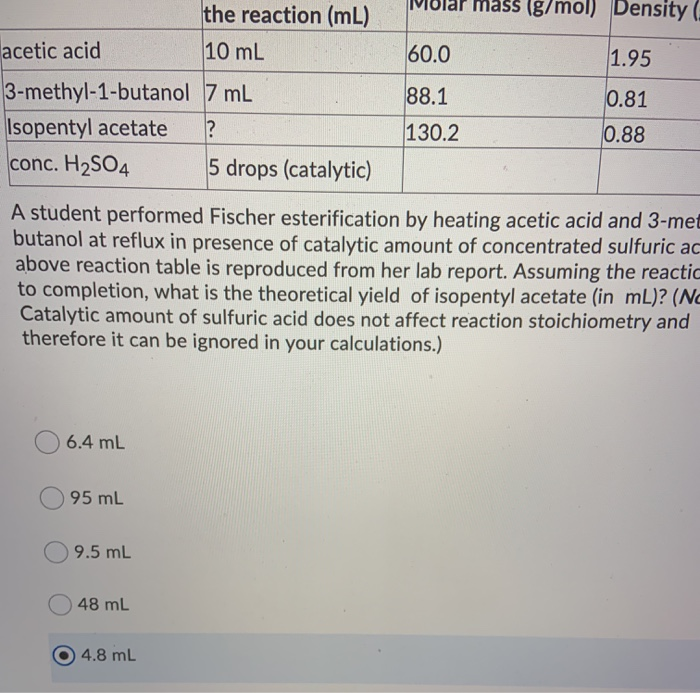
Mass of isopentyl alcohol= 1.0 mL × 0.809 g/mL = 0.809 grams isopentyl alcohol. So, the density of acetic acid = 1.05 g/mL and the density of isopentyl alcohol= 0.809 g/mL. molar mass of Acetic Acid mol Convert grams Acetic Acid to moles - Conversion of Measurement Units. => Things to know: The density of acetic acid = 1.05 g/cm^3 and the density of isopentyl alcohol= 0.809 g/cm^3, 1 mL = 1 cm^3, molar mass of isopentyl alcohol = 88.179 g/mol. Do a quick conversion: 1 grams Acetic Acid 0.016652245821785 mole using the molecular weight calculator and the molar mass of CH3COOH. We are given 1.0 mL of acetic acid and 1.0 mL of isopentyl alcohol. The equation (1) shows that one mole of acetic acid react with one mole of isopentyl alcohol to give one mole of isopentyl acetate and one mole of water. #"Mass %" = (0.5040 color(red)(cancel(color(black)("g"))))/(10.06 color(red)(cancel(color(black)("g")))) × 100 % = 5.The equation of of Reaction is given below ĬH3COOH + C5H11OH > CH3COOC5H11 + H2O. The following equation is used for calculating acid and base molarity where the concentration is given in wt : ( × d) / MW × 10 Molarity Where: Weight d Density (or specific gravity) MW Molecular Weight (or Formula Weight). We have 8.393 mmol of acetic acid in 10.00 mL of vinegar. #color(blue)(bar(ul(|color(white)(a/a)"Mass percent" = "mass of sample"/"mass of solution" × 100 %color(white)(a/a)|)))" "# Use the titration data to calculate the molarity.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
